Rb掺杂对K-Cs-Sb阴极材料光电性质的影响
Alkali antimonide photocathodes are widely used in many fields such as radiation detection, photon counting, and accelerator electron source due to their advantages of high quantum efficiency, long lifespan, short response time, and low preparation cost. Since K2CsSb bi-alkali photocathode has high photosensitivity ranging from 300 nm to 650 nm, it is often used as the key component of large-area microchannel plate photomultiplier tube and dynode photomultiplier tube. K-Cs-Rb-Sb tri-alkali photocathodes may exhibit more outstanding performance in spectral response enhancement and thermionic emission suppression compared to conventional K2CsSb bi-alkali photocathode. So far, there have been little theoretical researches on K-Cs-Rb-Sb tri-alkali photocathodes. Due to the difficulty in controlling the stoichiometric ratio of alkali metal elements during the actual preparation processes of K-Cs-Rb-Sb photocathodes, and in fact K-Cs-Rb-Sb tri-alkali photocathodes with different stoichiometric ratios have different photoemission properties, it is necessary to analyze the mechanism of Rb doping leading to different photocathode properties from the atomic and electronic perspective, thereby providing theoretical guidance for designing excellent alkali antimonide photocathodes.
The K2Cs2-xRbxSb bulk models and the (111)-oriented surface models with different Cs/Rb ratios corresponding to K2CsSb,K2Cs0.75Rb0.25Sb,K2Cs0.5Rb0.5Sb,K2Cs0.25Rb0.75Sb,and K2RbSb were established. The K2CsSb unit cell belongs to the DO3 cubic structure with a lattice constant of 0.8615 nm, and the space group is Fm-3m. According to the number of Cs atoms in K2CsSb replaced by Rb atoms, the lattice constants of several K-Cs-Rb-Sb bulk models after atom replacements were obtained by Vegard law. On the basis of the K2CsSb (111) Cs-terminated surface, six, eight, twelve, and sixteen Cs atoms were replaced from top to bottom, to obtain the K-Cs-Rb-Sb(111) surface models with different Cs/Rb ratios. To eliminate inter-layer interactions caused by the periodic mirror interaction between the surface slabs, a vacuum layer of 2 nm was set along the z-axis, including an upper vacuum layer with a thickness of 1.5 nm and a lower vacuum layer with a thickness of 0.5 nm. During the structural optimization process, the upper surface atoms with a thickness of 0.8 nm were allowed to fully relax, while the remaining atoms were constrained. The VASP software package using the first-principles calculation method based on the density functional theory was adopted. The projected augmented wave method was used as the pseudo potential, the generalized gradient approximation function proposed by Perdew-Burke-Ernzerhof was used to express the exchange correlation interaction, the plane wave expansion with a cut-off energy of 500 eV was used, and the conjugate gradient method was used to optimize the lattice constants and atom positions of the diverse models. The K-point grid in the Monkhorst-Pack form was set as 6×6×6 for bulk models and 6×6×1 for surface models, respectively.
The calculation results indicate that when Rb atoms replace Cs atoms in the K-Cs-Rb-Sb bulk models with different Cs/Rb ratios, the optical properties including reflectivity, refractive index, extinction coefficient, and absorption coefficient are hardly affected by Rb doping. This implies that the incorporation of Rb atoms has minimal impact on the optical properties of K2CsSb material. From the perspective of formation energy and formation enthalpy, all the K-Cs-Rb-Sb bulk models where Rb atoms replace K atoms have positive formation energies, and the corresponding formation enthalpies are larger than that of the K2CsSb model. This indicates that it is very difficult for K atoms to be replaced by Rb atoms in the preparation process of K-Cs-Rb-Sb tri-alkali photocathodes. At the same time, all K-Cs-Rb-Sb bulk models where Rb atoms replace Cs atoms have negative formation energies, and the corresponding formation enthalpies are less than that of the K2CsSb model, indicating that all the models where Rb atoms replace Cs atoms are easy to form with better thermodynamic stability. As the number of Rb atoms replacing Cs atoms increases, the formation energies and formation enthalpies gradually decrease. This means that in the presence of both Cs and Rb, the K2Cs0.25Rb0.75Sb model is the easiest to form and the most stable. All K-Cs-Rb-Sb bulk models exhibit the property of p-type semiconductor, and K2Cs0.25Rb0.75Sb has the smallest bandgap. For K-Cs-Rb-Sb surface models with different Cs/Rb ratios, the vacuum levels, surface energies, and electron effective masses gradually decrease. Among them, the K2Cs0.25Rb0.75Sb surface model has the smallest ionization energy, indicating that its electrons generated under external light excitation are more likely to transit from the valence band top to the conduction band bottom and move in the conduction band. This is beneficial for enhancing the spectral response of the photocathode and further improving the photoelectric conversion efficiency. Doping Rb element in K2CsSb can increase the work function of the surface model. On the whole, the K2CsRb0.250.75Sb (111) with a larger work function and surface can prevent the escape of some hot electrons while ensuring that a large number of photoelectrons can escape from the surface, in order to achieve the reduction of cathode dark current without reducing its quantum efficiency. In the surface model containing K, Cs, and Rb alkali metals, K2Cs0.25Rb0.75Sb has the highest conductivity, because the concentration of conduction band electrons gradually increases, and the effective mass of conduction band electrons in the surface model decreases as the number of Cs atoms replaced by Rb atoms increases.
When Rb atoms replace Cs atoms, Rb doping has little effect on the optical properties of K-Cs-Rb-Sb cathode materials. For K-Cs-Rb-Sb bulk models with different Cs/Rb ratios, K2Cs0.25Rb0.75Sb has the lower formation energy and formation enthalpy, indicating that it is easy to form under natural conditions and it is thermodynamically stable. For the surface models, K2Cs0.25Rb0.75Sb has the smaller surface energy and higher conductivity, as well as the smallest bandgap and ionization energy. Besides, the work function of K2Cs0.25Rb0.75Sb is larger than that of K2CsSb. Therefore, the K-Cs-Rb-Sb cathode with a Cs/Rb ratio (atomic number fraction) of 1∶3 is considered to be a stable photoemission material with high quantum efficiency, low dark current, and good conductivity. The research results can provide guidance for the preparation of high-performance K-Cs-Rb-Sb photocathodes. In the traditional K2CsSb photocathode preparation process, doping Rb elements can reduce the dark noise of the photomultiplier tube while maintaining a high level of quantum efficiency, thereby improving the detection sensitivity and accuracy of the device in practical applications.
1 引言
光电阴极是光电发射器件中完成光电转换的核心部件。碱锑化合物K-Cs-Sb光电阴极具有量子效率高、寿命长、响应时间短且制备成本低等优势[1],被广泛应用于辐射探测、光子计数和加速器电子源等领域[2-6]。传统的K-Cs-Sb光电阴极制备工艺较为简单:在真空环境下将Sb先蒸发在衬底上,随后依次引入或交替引入K、Cs与Sb反应,监测阴极光电流到达峰值[7-9]。研究者发现:相比于K-Cs-Sb光电阴极,掺Rb的K-Cs-Rb-Sb光电阴极在光谱响应提升和本征发射抑制方面更为突出[10]。典型的K-Cs-Rb-Sb光电阴极的制备工艺如下:首先在常温下蒸一层Sb,随后在180~200 ℃温度下蒸K;沉积第二层Sb,随后在180~200 ℃温度下蒸Rb;沉积第三层Sb,随后在160 ℃温度下蒸Cs;最后在室温下进行表面氧敏化,使灵敏度达到最高值[11]。此外,Cultrera等[10]采取不同工艺在蒸Rb的过程中待光电流不再增加时直接蒸Cs,制备的K-Cs-Rb-Sb阴极在可见光的量子效率得到明显提升,光谱响应向红外波段延伸。高鲁山等[12]以Na2KSb为基底层,然后用Cs、Rb、Sb进行表面处理,制备出具有高灵敏度、低热发射的四碱光电阴极[Rb,Cs]Na2KSb,其绿光到红光的光谱响应得到了明显改善;王宝林等[13]在此基础上提出了[Rb,Cs]Na2KSb光电阴极的表面能带结构模型,对实验结果进行了解释。
在K-Cs-Sb阴极材料理论研究方面,Ettema等[14]采用定域球面波方法对K2CsSb和KCs2Sb的电子结构进行了研究。Kalarasse等[15-16]采用全势线性化增强平面波法研究了不同K/Cs比例(原子数分数之比)的K-Cs-Sb化合物的电学和光学性质。舒昭鑫等[17]研究了不同反位、空位缺陷对K-Cs-Sb光电性质的影响。Wang等[18]研究了不同晶面和不同原子终止面的K2CsSb阴极的表面性质。Murtaza等[19]计算了不同双碱锑化物(Na2KSb、Na2RbSb、Na2CsSb、K2RbSb、K2CsSb和Rb2CsSb)的结构、电学和光学性质。然而,到目前为止,关于K-Cs-Rb-Sb阴极材料性质以及掺Rb的作用机理的理论研究还未见报道。在阴极制备过程中,碱金属元素的化学计量比难以控制,不同元素比例的光电阴极存在光电发射性能差异,因此有必要从原子电子层面研究元素化学计量比对K-Cs-Rb-Sb阴极性质的影响,以为研制更高性能的碱锑化合物阴极提供理论指导。
本文基于密度泛函理论(DFT)的第一性原理方法[20],采用VASP量子力学-分子动力学模拟软件包计算分析不同Cs/Rb比例下K-Cs-Rb-Sb体模型以及(111)表面模型的性质,得出Cs/Rb比例对电子结构和光学性质的影响规律,探索出最适合光电发射的K-Cs-Rb-Sb阴极材料。
2 理论模型与计算方法
为了研究不同Cs/Rb比例的K-Cs-Rb-Sb光电阴极材料性质,本文建立了K2Cs2-xRbxSb(x分别取0、0.25、0.5、0.75、1)的体模型以及与之相对应的(111)表面模型。K2CsSb晶胞属于晶格常数为0.8615 nm的DO3立方结构[17],所属空间群为Fm-3m[21]。用Rb原子替换K2CsSb中的Cs原子,根据Vegard定律计算出K2Cs2-xRbxSb的晶格常数[22],其表示式为
式中:a表示晶胞的晶格常数;x表示Rb在Cs、Rb总原子数中的占比;
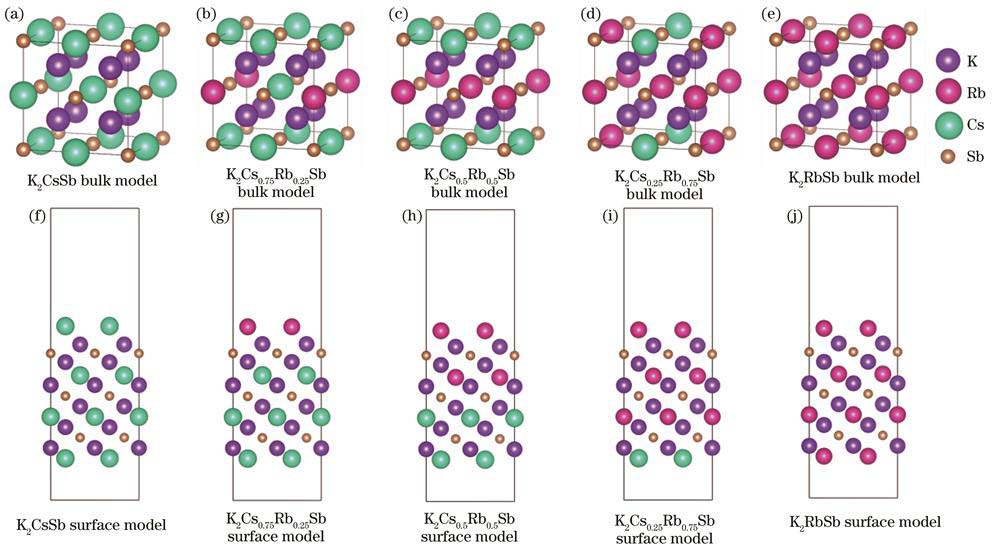
图 1. 不同Cs/Rb比例的K-Cs-Rb-Sb体模型和表面模型
Fig. 1. K-Cs-Rb-Sb bulk and surface models with different Cs/Rb ratios
K2CsSb光电阴极的(111)表面模型存在各种类型的周期性排列,如K/Cs/K/Sb、Cs/K/Sb/K、K/Sb/K/Cs和Sb/K/Cs/K。Wang等[18]在研究不同晶面K2CsSb模型性质中证明了富Cs的(111)表面具有最小的功函数,最利于光电发射,因此本文以富Cs的K2CsSb(111)表面为研究对象,从上至下分别替换4、8、12、16个Cs原子,得到不同Cs/Rb比例的K-Cs-Rb-Sb(111)表面模型,如
本文计算使用VASP软件包中的投影缀加波(PAW)作为赝势,采用基于Perdew-Burke-Ernzerhof(PBE)函数的广义梯度近似法(GGA)分析电子交换-关联作用。平面波展开的截断能为500 eV,高斯展宽为0.05 eV,采用共轭梯度法来优化模型的晶格常数和原子位置。总能量收敛标准为1×10-6 eV,原子间的相互作用受力小于0.01 eV/Å。对于体模型,用于布里渊区采样的Monkhorst-Pack形式的K点网格是6×6×6;而对于表面模型,这些网格被设定为6×6×1,所有的计算均在倒空间里进行。参与计算的赝势原子的元素价电子态为K:3s23p64s1、Rb:4s24p65s1、Cs:5s25p66s1和Sb:5s25p3[17]。
3 结果与讨论
3.1 K-Cs-Rb-Sb体模型性质分析
在K2CsSb结构中掺Rb,形成K-Cs-Rb-Sb四元化合物结构的难易程度可以用形成能表示,其计算公式[17]如下:
式中:
形成焓可以表示物质形成的热力学稳定性,其值越小表示物质在热力学上越稳定。对于不同组成的K2Cs2-xRbxSb体模型,其形成焓
式中:
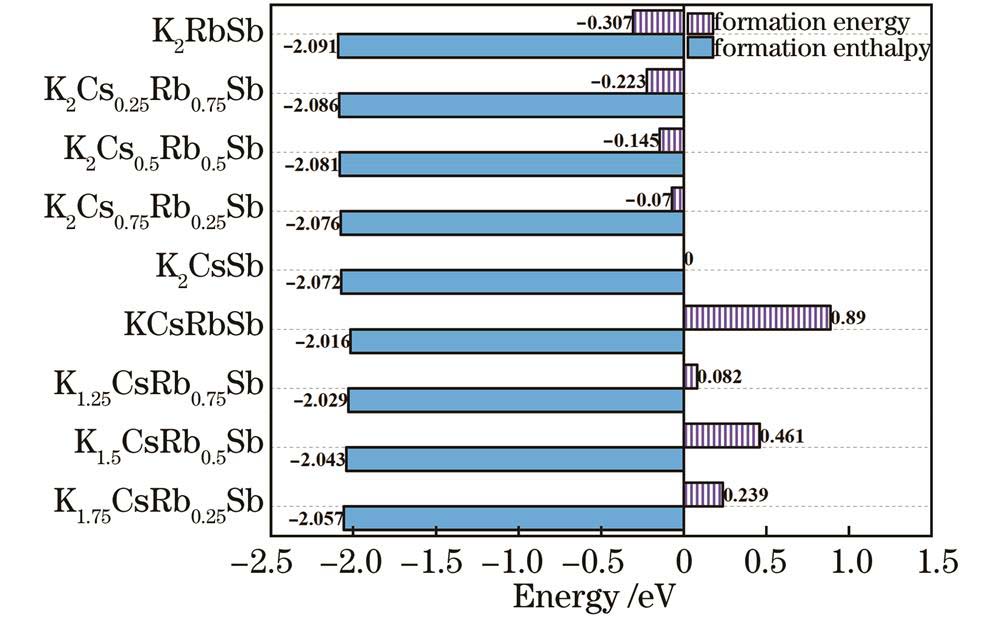
图 2. 不同Cs/Rb比例K-Cs-Rb-Sb体模型的形成能和形成焓
Fig. 2. Formation energy and formation enthalpy of K-Cs-Rb-Sb bulk models with different Cs/Rb ratios
光学性质作为K-Cs-Rb-Sb材料在光电阴极应用中的重要性质,包括反射率、折射率、消光系数和吸收系数,它们由介电常数推导得到,介电常数由实部
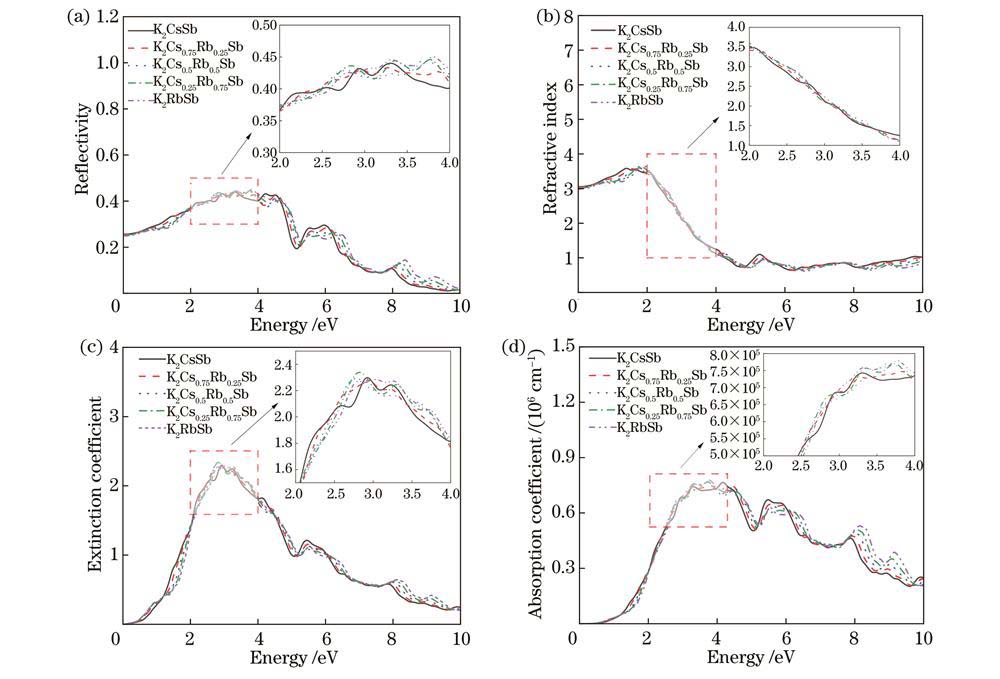
图 3. 不同Cs/Rb比例K-Cs-Rb-Sb体模型的光学性质。(a)反射率;(b)折射率;(c)消光系数;(d)吸收系数
Fig. 3. Optical properties of K-Cs-Rb-Sb bulk models with different Cs/Rb ratios. (a) Reflectivity; (b) refractive index; (c) extinction coefficient; (d) absorption coefficient
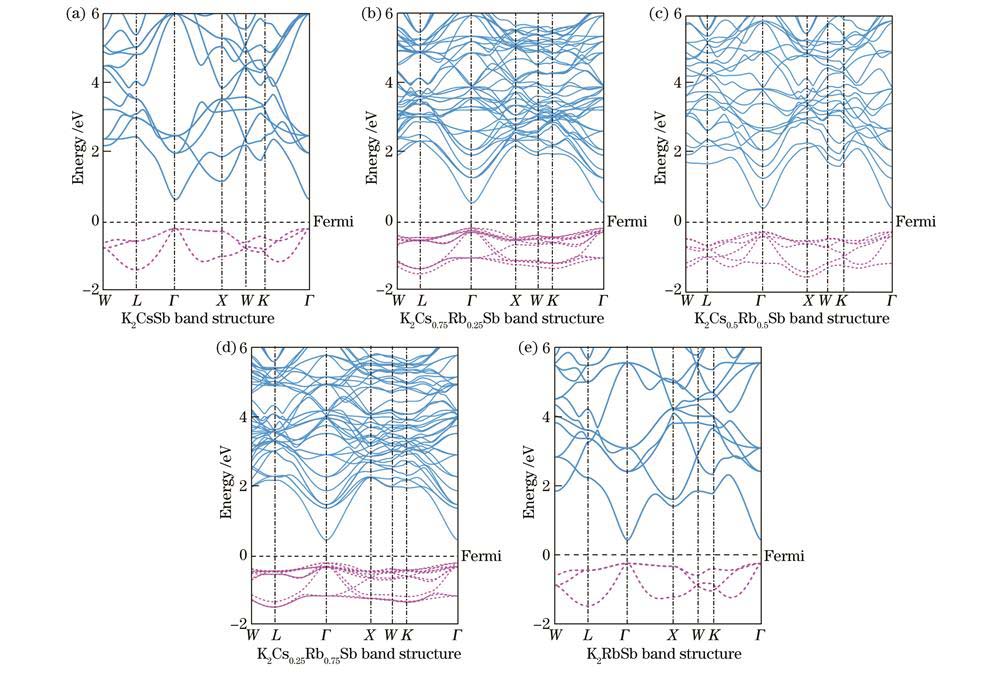
图 4. 不同Cs/Rb比例K-Cs-Rb-Sb体模型的能带
Fig. 4. Band structures of K-Cs-Rb-Sb bulk models with different Cs/Rb ratios
表 1. 不同Cs/Rb比例K-Cs-Rb-Sb体模型的禁带宽度
Table 1. Band gaps of K-Cs-Rb-Sb bulk models with different Cs/Rb ratios
|
3.2 K‑Cs‑Rb‑Sb表面模型性质分析
半导体的功函数表征了光电子从表面逃逸到真空中的能力,是光电发射“三步模型”理论中的重要参数[24],可表示为
式中:
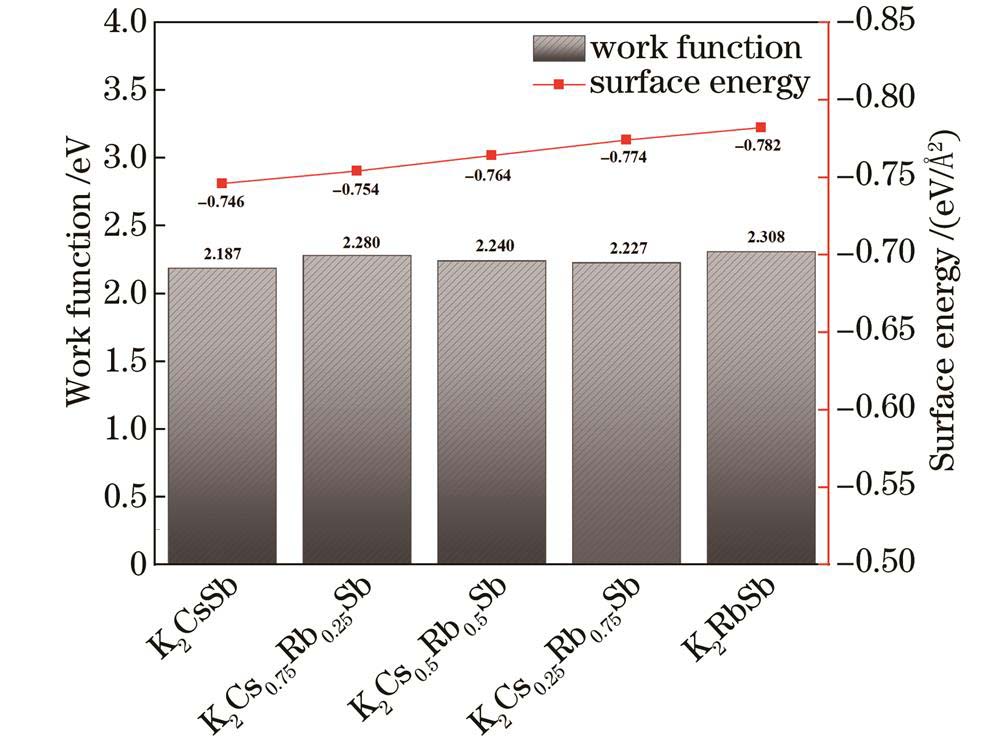
图 5. 不同Cs/Rb比例K-Cs-Rb-Sb(111)表面模型的功函数和表面能
Fig. 5. Work function and surface energy of K-Cs-Rb-Sb (111) surface models with different Cs/Rb ratios
对于光电阴极器件而言,当探测微弱信号时,热发射形成的暗电流会对微弱信号的探测产生较大干扰,暗电流越小则光电倍增管器件对于极微弱信号的探测越有优势;量子效率代表了光电阴极进行光电转换的能力,是指光电阴极在接收到光子后成功产生并释放电子的概率,量子效率高意味着光电阴极对光子的响应更灵敏,输出信号更强。暗电流和量子效率这两项重要指标都与功函数有很大的关系。首先,热电子发射电流与热力学温度和功函数相关,在温度相同的情况下,功函数的大小决定了阴极面的热电子发射量,功函数的降低会导致热电子发射量变大,然而功函数升高也会导致光电阴极器件的量子效率变低。综合来看,Cs∶Rb为1∶3的K-Cs-Rb-Sb(111)表面可以在保证大量光电子能够逸出表面的前提下,阻止部分热电子逸出,以达到在降低阴极暗电流的同时不降低量子效率的目的。
表面能
式中:Eslab表示已经完全弛豫的表面模型的总能量;Ebulk表示K-Cs-Rb-Sb体模型的总能量;n表示该表面结构中体模型的单元数;2A表示上下表面积之和。由于(111)表面具有冗余原子,本文将计算公式改为以下公式来近似得到表面能:
式中:nK、nCs、nRb、nSb分别为K、Cs、Rb、Sb的原子数目;μK、μCs、μRb、μSb分别为K、Cs、Rb、Sb原子的化学势。通过计算得到5种Cs/Rb比例下阴极(111)表面的表面能如
半导体的离化能代表电子从价带顶跃迁到真空能级的能力。离化能I[26]可表示为
式中:
表 2. 不同Cs/Rb比例K-Cs-Rb-Sb(111)表面模型的真空能级、价带顶能量和离化能
Table 2. Vacuum level, valence band top energy, and ionization energy of K-Cs-Rb-Sb (111) surface models with different Cs/Rb ratios
|
电导率σ反映了半导体的导电能力,根据Drude模型,其表达式[27]为
式中:q为电子电荷量;n为载流子浓度;μ为载流子迁移率,表征载流子在电场作用下加速运动快慢的一个物理量,其表达式[28]为
式中:t为散射时间;meff为电子有效质量。根据能带结构,导带的电子有效质量meff[21]可以表示为
式中:
表 3. 不同Cs/Rb比例K-Cs-Rb-Sb(111)表面模型的电子有效质量
Table 3. Electron effective mass of K-Cs-Rb-Sb (111) surface models with different Cs/Rb ratios
|
此外,增大载流子浓度也有利于提高电导率。表面模型中载流子浓度主要由导带电子贡献,导带电子浓度n[18]可以表示为
式中:V为表面模型体积;DC(E)为导带附近的态密度;f (E)为电子的费米-狄拉克分布。若阴极材料呈现p形半导体、n型表面态性质,则可以获得较好的光电发射能力。根据表面模型能带结构,发现K2CsSb、K2Cs0.75Rb0.25Sb、K2Cs0.5Rb0.5Sb、K2Cs0.25Rb0.75Sb、K2RbSb这5种表面模型呈现n型表面性质,计算得到的导带电子浓度分别为7.944×1018 cm-3、7.255×1018 cm-3、7.031×1019 cm-3、7.336×1019 cm-3、7.795×1019 cm-3。可以看出,在含有K、Cs、Rb 3种碱金属的表面模型中,随着Rb原子替换Cs的原子数目的增加,表面模型的导带电子浓度逐渐增加,K2Cs0.25Rb0.75Sb的导带电子有效质量最小。根据
4 结论
本文分别建立了具有DO3立方结构的K2CsSb、K2Cs0.75Rb0.25Sb、K2Cs0.5Rb0.5Sb、K2Cs0.25Rb0.75Sb、K2RbSb 5种碱锑化合物阴极材料的体模型以及与其相对应的(111)表面模型,并采用基于密度泛函理论的第一性原理方法计算了其电子结构和光学性质。计算结果表明,当Rb原子替换Cs原子掺杂时,Rb掺杂对K-Cs-Rb-Sb阴极材料的反射率、折射率、消光系数等光学性质影响甚微。对于不同Cs/Rb比例的K-Cs-Rb-Sb体模型而言,Cs/Rb比例为1∶3的K2Cs0.25Rb0.75Sb具有较低的形成能和形成焓,在自然条件下容易形成且热力学稳定。对于表面模型而言,K2Cs0.25Rb0.75Sb具有较小的表面能、较小的电子有效质量以及较高的电子浓度,还具有最小的禁带宽度和最低的离化能;由于K2Cs0.25Rb0.75Sb的功函数大于K2CsSb,因此,Cs/Rb比例为1∶3的K-Cs-Rb-Sb阴极被认为是具有量子效率高、暗电流低且导电性好等优点的稳定光电发射材料。研究结果对制备高性能的K-Cs-Rb-Sb光电阴极具有一定的指导意义,在传统K2CsSb光电阴极制备过程中,通过掺杂Rb元素使光电倍增管在保持量子效率较高水平的基础上降低暗噪声,从而提升器件在实际应用中的探测灵敏度和准确性。
[1] 安迎波. 碱锑光电阴极特性模拟研究[D]. 西安: 中国科学院西安光学精密机械研究所, 2014.
AnY B. Simulation study on characteristics of alkali antimony photocathode[D]. Xi'an: Xi'an Institute of Optics and Precision Mechanics, Chinese Academy of Sciences, 2014.
[2] Ren L, Sun J N, Si S G, et al. Study on the improvement of the 20-inch microchannel plate photomultiplier tubes for neutrino detector[J]. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment, 2020, 977: 164333.
[3] 曾雨珊, 余谢秋, 田野. 从太赫兹波到光波驱动的集成电子加速器研究进展[J]. 中国激光, 2023, 50(17): 1714008.
[4] 王爱伟, 李驰, 戴庆. 基于相干电子源的超快低能电子全息成像[J]. 中国激光, 2023, 50(1): 0113003.
[5] Xie J Q, Attenkofer K, Demarteau M, et al. Large area planar photocathode for MCP-based photodetectors[J]. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment, 2020, 955: 163234.
[6] Dunham B, Barley J, Bartnik A, et al. Record high-average current from a high-brightness photoinjector[J]. Applied Physics Letters, 2013, 102(3): 034105.
[7] Ghosh C, Varma B P. Preparation and study of properties of a few alkali antimonide photocathodes[J]. Journal of Applied Physics, 1978, 49(8): 4549-4553.
[8] Xie J Q, Demarteau M, Wagner R, et al. Synchrotron X-ray study of a low roughness and high efficiency K2CsSb photocathode during film growth[J]. Journal of Physics D: Applied Physics, 2017, 50(20): 205303.
[9] Zhang F, Li X P, Li X S. Development of preparation systems with K2CsSb photocathodes and study on the preparation process[J]. Chinese Physics Letters, 2019, 36(2): 022901.
[10] Cultrera L, Gulliford C, Bartnik A, et al. Rb based alkali antimonide high quantum efficiency photocathodes for bright electron beam sources and photon detection applications[J]. Journal of Applied Physics, 2017, 121(5): 055306.
[11] DvořákM. Some properties of the trialkali Sb-K-Rb-Cs photocathode[M]//Photo-electronic image devices, proceedings of the fourth symposium. Amsterdam: Elsevier, 1969: 347-355.
[12] 高鲁山, 李朝木. 四碱光阴极的研究[J]. 电子学报, 1986, 14(5): 27-31.
Gao L S, Li C M. Investigation of multi-alkali photocathode[J]. Acta Electronica Sinica, 1986, 14(5): 27-31.
[13] 王宝林, 李朝木, 曾正清, 等. 改进型[Cs, Rb]Na2KSb光阴极的制备研究[J]. 真空与低温, 2009, 15(2): 112-116.
Wang B L, Li C M, Zeng Z Q, et al. Preparation and study of improvement [Cs, Rb]Na2KSb photo cathode[J]. Vacuum and Cryogenics, 2009, 15(2): 112-116.
[14] Ettema A R H F, de Groot R A. Electronic structure of Cs2KSb and K2CsSb[J]. Physical Review B, 2002, 66(11): 115102.
[15] Kalarasse L, Bennecer B, Kalarasse F. Optical properties of the alkali antimonide semiconductors Cs3Sb, Cs2KSb, CsK2Sb and K3Sb[J]. Journal of Physics and Chemistry of Solids, 2010, 71(3): 314-322.
[16] Kalarasse L, Bennecer B, Kalarasse F, et al. Pressure effect on the electronic and optical properties of the alkali antimonide semiconductors Cs3Sb, KCs2Sb, CsK2Sb and K3Sb∶Ab initio study[J]. Journal of Physics and Chemistry of Solids, 2010, 71(12): 1732-1741.
[17] 舒昭鑫, 张益军, 王兴超, 等. 反位缺陷对K2CsSb光阴极光电性质的影响[J]. 光学学报, 2021, 41(12): 1216001.
[18] Wang X C, Zhang K M, Jin M C, et al. First-principles investigation of structural, electronic and optical properties of cubic K2CsSb with different surface orientations[J]. Solid State Communications, 2022, 356: 114960.
[19] Murtaza G, Ullah M, Ullah N, et al. Structural, elastic, electronic and optical properties of bi-alkali antimonides[J]. Bulletin of Materials Science, 2016, 39(6): 1581-1591.
[20] Hafner J. Ab-initio simulations of materials using VASP: density-functional theory and beyond[J]. Journal of Computational Chemistry, 2008, 29(13): 2044-2078.
[21] Shabaev A, Jensen K L, Finkenstadt D, et al. Density of states of Cs3Sb calculated using density-functional theory for modeling photoemission[J]. Proceedings of SPIE, 2017, 10374: 103740L.
[22] Linnik M, Christou A. Calculations of optical properties for quaternary III-V semiconductor alloys in the transparent region and above (0.2-4.0 eV)[J]. Physica B: Condensed Matter, 2002, 318(2/3): 140-161.
[23] 张登琪, 田汉民, 何全民, 等. 低浓度Ge掺杂及Sn、Ge替换CsPbI3的电光学性质[J]. 激光与光电子学进展, 2023, 60(15): 1516002.
[24] Bechstedt F, Scheffler M. Alkali adsorption on GaAs(110): atomic structure, electronic states and surface dipoles[J]. Surface Science Reports, 1993, 18(5/6): 145-198.
[25] Lang N D, Kohn W. Theory of metal surfaces: charge density and surface energy[J]. Physical Review B, 1970, 1(12): 4555-4568.
[26] 吴孔平, 孙昌旭, 马文飞, 等. 铝-金刚石界面电子特性与界面肖特基势垒的杂化密度泛函理论HSE06的研究[J]. 物理学报, 2017, 66(8): 088102.
Wu K P, Sun C X, Ma W F, et al. Interface electronic structure and the Schottky barrier at Al-diamond interface: hybrid density functional theory HSE06 investigation[J]. Acta Physica Sinica, 2017, 66(8): 088102.
[27] Phuc H V, Hieu N N, Hoi B D, et al. Tuning the electronic properties, effective mass and carrier mobility of MoS2 monolayer by strain engineering: first-principle calculations[J]. Journal of Electronic Materials, 2018, 47(1): 730-736.
[28] Künzel H, Döhler G H, Ploog K. Determination of photoexcited carrier concentration and mobility in GaAs doping superlattices by Hall effect measurements[J]. Applied Physics A, 1982, 27(1): 1-10.
韩允锋, 金睦淳, 任玲, 王兴超, 张锴珉, 刘晓荣, 钱芸生, 张益军. Rb掺杂对K-Cs-Sb阴极材料光电性质的影响[J]. 光学学报, 2024, 44(4): 0416001. Yunfeng Han, Muchun Jin, Ling Ren, Xingchao Wang, Kaimin Zhang, Xiaorong Liu, Yunsheng Qian, Yijun Zhang. Effect of Rb Doping on Photoelectric Properties of K-Cs-Sb Cathode Material[J]. Acta Optica Sinica, 2024, 44(4): 0416001.






