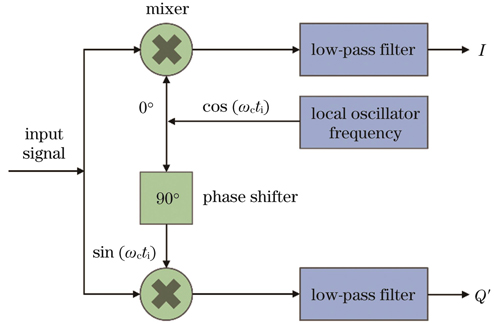
基于激光外差干涉技术的非接触式光声信号检测研究【增强内容出版】
Photoacoustic imaging is a non-invasive, functional optical imaging technique that uses the photoacoustic effect with ultrasound as a mediator. Despite its potential for various medical applications, traditional contact-based signal detection methods have hindered its progress in clinical practice. To overcome these limitations, non-contact detection of photoacoustic signals has emerged as a solution. This approach uses air-coupled or all-optical detection methods and achieves a wide bandwidth and high sensitivity for ultrasound signal reception, aligning with the demands of modern medical technology. Current contact-based photoacoustic imaging still faces challenges, such as signal attenuation, surface scattering, system sensitivity, and environmental interference. Therefore, in this study, a noncontact, noninvasive, and cost-effective photoacoustic signal detection device, based on the optical heterodyne technology, is proposed. We aim to address the challenges in photoacoustic signal detection, reduce system costs and complexity, and promote the practical application of non-contact photoacoustic imaging in clinical settings.
The photoacoustic signals are detected using heterodyne interferometry. First, based on the characteristic parameters of tissue surface vibration signals and the propagation properties of ultrasound waves in a medium, the relationship between the vibration displacement and pressure causing the vibrations is established. Subsequently, by employing the dual-frequency optical heterodyne interferometric theory, the relationship between the photoacoustic signal-induced tissue surface vibration displacement and phase changes is established. Furthermore, I/Q quadrature demodulation is applied to process the photoacoustic signals; this ensures system stability and high demodulation accuracy in complex environments. To achieve all-optical detection of photoacoustic signals, an photoacoustic signal detection system is designed using a He-Ne laser as the detection light source. The optical components are configured to construct an optical-path system for precise detection of photoacoustic signals. In the experiments, an electro-optic Q-switched dual-wavelength Nd∶YAG laser is selected as the excitation light source. This laser allows for flexible control of the excitation light conditions by adjusting the beam diameter and intensity. Finally, the acoustic vibration characteristics induced in the sample under stimulation are evaluated by observing the phase changes and vibration displacement of the photoacoustic signals.
In this study, we investigate the detection of ultrasound vibrations generated by an ultrasound transducer (UST) using a photoacoustic signal full-optical detection system and an immersion probe. A comparative analysis of the measurement results reveals that the heterodyne interference system effectively reconstructs the ultrasound vibrations. The demodulated ultrasound displacement pulse envelope is closely aligned with the measurement result obtained using the immersion probe (Figs.3 and 4). Under constant input parameters, varying the amplitude of the power supply voltage results in a linear increase in the ultrasound signal intensity, which is measured by both the heterodyne system and immersion probe (Fig.5). Furthermore, different driving frequencies are set with constant input signal parameters. The ultrasound vibration frequencies measured by our system and the immersion probe are comparatively analyzed. According to the analysis results, the I/Q quadrature demodulation method accurately extracts ultrasound vibration frequencies with minimal relative measurement error, exhibiting an absolute difference of 0.2 kHz (Table 1). In the absence of excitation pulses, the demodulated baseline interference signal displays subtle displacement deviations with displacement offset control at the nanometer level (Fig.8). The detection of ultrasound vibrations on the surface of a carbon rod demonstrates a close agreement between the ultrasound vibration frequencies demodulated by our system and those measured by the immersion probe (Figs.6 and 7). However, in the case of ultrasound detection on the surface of the porcine liver, the rigidity of measurement mirror M2 leads to higher-frequency vibrations demodulated by our system compared to those measured by the immersion probe (Figs.9 and 10).
In this study, optical heterodyne interferometry and digital Doppler signal demodulation methods are used to experimentally investigate non-contact photoacoustic signal detection. The acoustic vibration characteristics of the target are extracted through the demodulation of the dual-frequency optical heterodyne interference signals. Subsequently, an ultrasonic transducer is employed to simulate the high-frequency vibration signals, and a performance test is conducted. The experimental results demonstrate that the heterodyne system effectively reconstructs photoacoustic vibration signals with minimal relative frequency deviation compared to the immersion probe. In addition, experiments involving a carbon rod and excised biological tissue reveal that the heterodyne system based on the I/Q orthogonal method exhibits superior performance in demodulating photoacoustic signals owing to its insensitivity to changes in the interference signal amplitude. Moreover, the designed photoacoustic signal detection system incorporates linearly polarized light and heterodyne interferometry and the detection sensitivity and precision are enhanced. The heterodyne optical path structure, which is easily adjustable and effective in suppressing interference noise, contributes to the robust performance of the system. By utilizing high-speed data acquisition cards and digital signal processing techniques for interference signal processing, the demodulation algorithm proves to be simple and flexible in design, offering potential cost savings in hardware implementation. The research findings suggest that the proposed system holds a certain reference value for clinical applications in noncontact photoacoustic signal detection.
1 引言
光声成像(PAI)是一种以超声为媒介的基于光声效应的无损功能性光学成像技术[1]。它利用了光学和超声的成像特性,以高对比度和良好的穿透性显示光吸收,可用于血氧饱和度成像、脑功能成像、基因表达、腔体病变诊断、皮肤黑色素瘤和纵向肿瘤血管生成以及蛋白质相互作用的成像和检测等[2-4]。PAI系统主要由两个部分组成:脉冲激光对组织吸收体的光学激发和超声传感器的声信号探测,后者在最终成像中起着至关重要的作用。传统接触式的超声探头需要与样品物理接触才能获得光声信号,这限制了其在临床和临床前应用中的灵活性[5]。为了克服局限性,光声信号的非接触探测方式应运而生,该方法使用空气耦合或全光检测的方法替代传统接触式超声传感器,实现了宽频带和高灵敏的超声信号接收,符合现代医学技术的发展需求。
在采用空气耦合的光声信号探测方法中,空气和被测样品之间的阻抗失配会导致超声信号严重衰减,且该方法系统的分辨率较低,较长的信号采样时间限制了其潜在的体内应用[6-7]。光学超声检测方法与传统的压电器件相比,具有更高的灵敏度和更宽的频带,也为开发小型化和光学透明的超声检测器提供了机会。当相干光照射发生机械形变的粗糙表面时,其后向散射光发生干涉,形成激光散斑,通过对这些散斑图样进行处理可以提取关于表面形变以及声压的信息。Li等[8]评估了使用双光束光学设计和互补金属氧化物半导体(CMOS)相机来描述光声信号强度的可行性,并将散斑图样作为时间函数进行关联,量化了光声扰动的强度。实验结果证明,相机的带宽不足以捕捉显著的MHz信号,且由于任何形式的运动和机械噪声都会扭曲探测光束的散斑图样,故该方法对相位噪声和环境扰动非常敏感,体内的应用将受到极大的限制。
光学相干测量法利用干涉仪从样品表面返回的光中提取相位对比度,从而克服散斑检测带来的一些问题。目前零差和外差结构都已被用于超声信号的接收。Lu等[9]使用相干长度约为17 mm的零差干涉仪,通过同步方法将系统锁定在最高灵敏度,实现了小鼠耳部血管的准非接触成像。Wang等[10]设计了一种基于3×3光纤耦合器的零差干涉仪(相干长度超过几百mm),该多端口干涉系统消除了初始相位和环境相位扰动的影响,并利用探针束的强度变化重建了光声信号。2015年,Eom等[11]设计了一种观察鸡胚绒毛尿囊膜(CAM)的光纤外差干涉仪,获得的血管结构的横向和轴向分辨率分别为100 μm和30 μm,可识别深度为2.5 mm。随后,Eom等[12]又将光声检测与用于内窥镜成像的光学相干断层扫描(OCT)相结合,进一步将系统设计成双模态结构。然而,外差干涉法很难在粗糙表面上探测到高度散射的反射光束,因此需要在样品表面上覆盖一层薄薄的水或油。
光声遥感(PARS)显微技术首次由Bell等[13]于2017年提出,用于光声信号的非接触式、非干涉式检测。目前,光声遥感技术在系统信噪比和分辨率、成像深度和对比度、扫描速度以及多模态应用等多方面取得了迅速发展。研究人员基于光声非接触探测机理,建立了一系列物理模型,得到了基于弹光效应的光声信号远程探测的仿真结果[14-15]。但PARS也存在一定的局限性,即由于是共聚焦配置,时间分辨信号不显示深度分辨信息。
由以上论述可知,基于不同原理的光声信号远程探测方法在近几年的实验研究中取得了很大的进展,然而目前非接触探测技术还存在一定的局限,尚无兼具各项最佳性能的信号探测方式,且该技术还处于临床实践前的实验研究阶段。因此本文设计了一种基于光外差技术的用于光声信号探测的非接触、无创以及低成本的检测装置,致力于解决光声信号探测中存在的难题,降低系统成本及复杂度,推进未来非接触式光声成像的临床实践。
2 原理与方法
2.1 光外差技术探测光声信号的原理
当生物组织受到光子的照射时,组织内部的目标生色团吸收光能,导致局部瞬态温升,引起局部压力的升高,信号向组织表面传播,由超声换能器接收信号。重建的信号可映射组织内部的原始光能沉积。当同时满足热限定和压力限定条件时,热膨胀会导致声压产生,即光声信号[16]:
式中:
式中:
由双频光外差干涉理论[20]可知,光声信号引起的组织体表面振动位移
式中:
通过光干涉信号相位的改变可计算出光声压的大小。对于已采集的干涉调相信号
式中:
采用相位展开法获取干涉信号的相位,被测样品表面的瞬时振动位移可表示为
式中:
将I/Q正交解调法用于解调光声信号,其抗干扰性强,对系统变化不敏感,可确保系统在复杂环境中的稳定性,高解调精度使其能准确提取光声信号的频谱信息。这些优势使得I/Q正交解调法成为非接触式光声信号探测中可靠的信号处理手段,为系统性能提升和实际应用奠定了基础。
2.2 光声信号全光探测系统
光声信号全光探测系统的原理图如
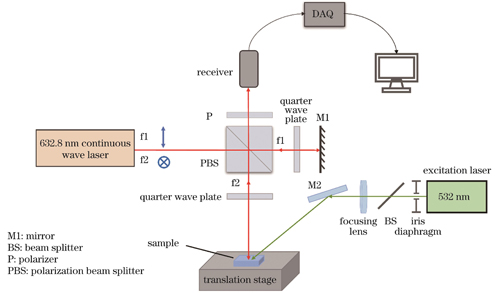
图 2. 光声信号全光探测系统的原理图
Fig. 2. Schematic of all-optical detection system for photoacoustic signals
实验选用电光调Q双波长Nd∶YAG激光器作为系统的激励光源,激发光源的工作波长为1064 nm/532 nm,单脉冲宽度约为7 ns,激光脉冲的重复频率可调范围为1~10 Hz,激光输出单脉冲能量调节范围为100~1000 mJ(波长为1064 nm时)和50~500 mJ(波长为532 nm时)。当系统工作时,激光束由导光臂出射,进入到激发光路。系统的激发光路主要由可变光阑、分光片以及聚焦透镜组成。分光片(BS)主要是将部分激发光耦合到PIN型光电探测器中,该激发光信号作为触发信号被反馈给采集卡(DAQ),用于控制采集卡和激发激光的同步触发信号采集。可变光阑主要用于调节光束直径,其可变孔径范围为
3 结果
3.1 模拟振动信号探测实验
为装调方便,实验时将干涉仪测量臂中的反射镜M2紧贴超声换能器(UST)一起放置于水槽中,信号发生器输出的方波信号驱动换能器发出超声波,该超声波被耦合至测量镜M2,发生同频正弦振动(欠阻尼的受迫振动)。将共振频率为500 kHz的压电陶瓷作为研制的聚焦超声换能器的压电元件,通过功率放大器将信号发生器的信号放大后施加在压电晶体上,压电晶体形变形成超声波。微型功率放大器的作用是提供足够大的输出功率以驱动换能器,从而得到实验所需的声压强度,超声换能器的驱动电压(即放大器输出电压)幅度取决于电源电压的大小,故实验中可以通过改变电源电压的幅度来获取不同强度的超声信号。经测试,电源电压-声压幅度曲线呈近似线性,且电源电压的理想工作范围为6~20 V,对应的声压强度
使用光声信号全光探测系统和水浸探头先后对UST产生的超声振动进行检测,且在相同实验条件下,每种方法各得到10次测量结果。实验中超声探头以水为耦合介质,被固定在距离测量镜M2表面约5 mm处,保证检测点与探测激光束指向的位置相同,采集到的信号由超声探头与M2耦合振动产生,水浸探头检测到的超声信号经超声前置放大器放大后被DAQ接收并保存于计算机。光声信号探测系统解调得到的是时域振动位移信号,通过傅里叶频谱分析可得到超声振动频率;水浸探头得到的是时域超声压电信号(结果为电压幅值,未进行声压标定),对其进行傅里叶频谱分析,同样可得到超声振动频率。
使用信号发生器驱动UST产生超声振动,设置频率为500 kHz、宽度为300 µs的脉冲方波为输入信号,电源电压设为10 V。DAQ的数据采集模式设置为单通道有限点采集(数字触发),设置采样率为200 MSa/s。由外差干涉系统探测并由I/Q正交法解调得到的时域位移信号及其时间轴局部展开图如
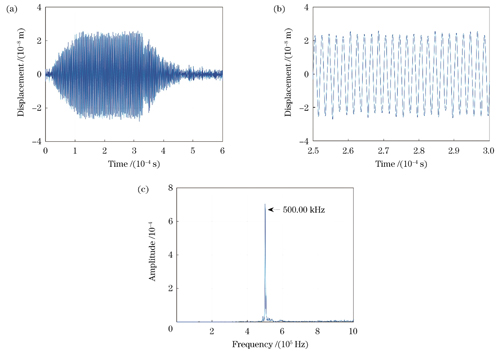
图 3. 由外差干涉系统探测并由I/Q正交法解调得到的超声振动信号。(a)时域位移信号;(b)位移信号展开图;(c)位移信号频谱图
Fig. 3. Ultrasonic vibration signal detected by heterodyne interference system and demodulated by I/Q orthogonal method. (a) Time-domain displacement signal; (b) unfolded diagram of displacement signal; (c) spectrogram of displacement signal
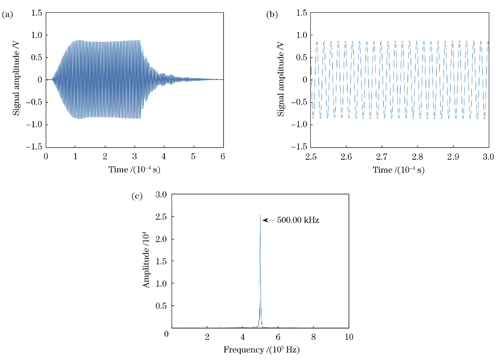
图 4. 超声探头探测到的超声压电信号。(a)时域超声信号;(b)超声信号展开图;(c)超声信号频谱图
Fig. 4. Ultrasonic piezoelectric signal detected by ultrasonic probe. (a) Time-domain ultrasonic signal; (b) unfolded diagram of ultrasonic signal; (c) spectrogram of ultrasonic signal
在输入信号参数不变的情况下,改变电源电压幅值,当电源电压从6 V增至20 V时,光声信号探测系统测得的超声振动位移幅值与水浸探头测得的超声信号电压幅值随电源电压的变化如
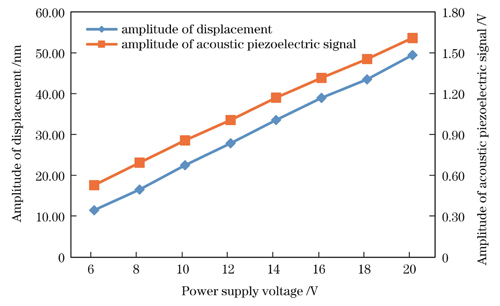
图 5. 位移信号幅值和声压电信号幅值随电源电压的变化
Fig. 5. Variation of amplitude of displacement signal and amplitude of acoustic piezoelectric signal with power supply voltage
在输入信号其他参数不变的情况下,设置驱动频率分别为500、525、550、575、600 kHz,电源电压保持10 V不变。
表 1. 外差系统和水浸探头检测到的超声振动信号结果的对比
Table 1. Comparison of results of ultrasonic vibration signals detected by heterodyne system and water immersion probe
|
3.2 光声信号探测实验
根据
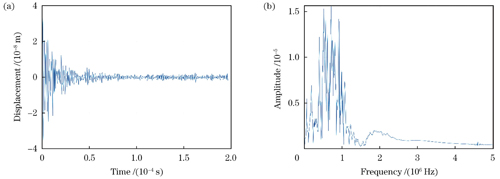
图 6. 由外差干涉系统探测并由I/Q正交法解调得到的碳棒表面的超声振动位移信号。(a)时域图;(b)频谱图
Fig. 6. Ultrasonic vibration displacement signal on surface of carbon rod detected by heterodyne interference system and demodulated by I/Q orthogonal method. (a) Time domain diagram; (b) spectrogram
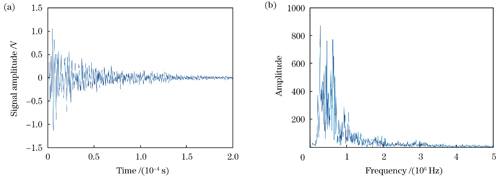
图 7. 水浸探头检测到的碳棒表面的光声信号。(a)时域图;(b)频谱图
Fig. 7. Photoacoustic signal on surface of carbon rod detected by water immersion probe. (a) Time domain diagram; (b) spectrogram
为验证系统的稳定性,在未施加激励脉冲时,利用基线干涉信号解调出时域振动位移偏差。实验结果如
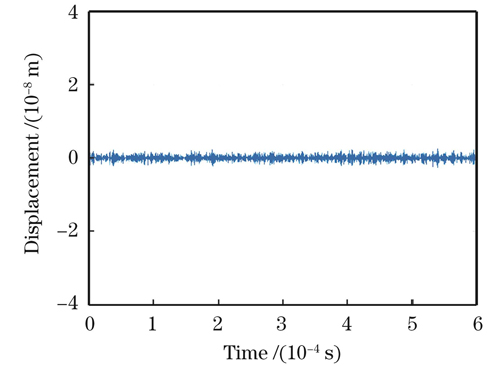
图 8. 无激励脉冲时I/Q正交法解调得到的时域振动位移信号
Fig. 8. Time-domain vibration displacement signal demodulated by I/Q orthogonal method without excitation pulse
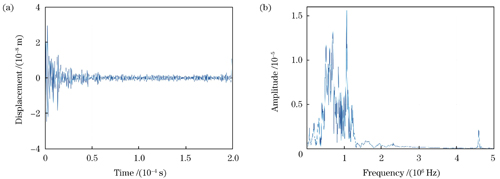
图 9. 由外差干涉系统探测并由I/Q正交法解调得到的猪肝表面的超声振动位移信号。(a)时域图;(b)频谱图
Fig. 9. Ultrasonic vibration displacement signal on porcine liver surface detected by heterodyne interference system and demodulated by I/Q orthogonal method. (a) Time domain diagram; (b) spectrogram
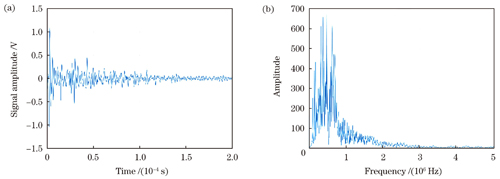
图 10. 水浸探头检测到的猪肝表面的光声信号。(a)时域图;(b)频谱图
Fig. 10. Photoacoustic signal on porcine liver surface detected by water immersion probe. (a) Time domain diagram; (b) spectrogram
4 结论
光声技术以其高光学对比度、较好的空间分辨率和非侵入性的特点在生物医学领域中得到了广泛应用。然而,传统的光声系统在获取光声信号时需要样品与换能器本身或耦合介质进行物理接触,这种接触式信号探测方式显然制约了其在临床中的应用。为克服压电式换能器的局限性,采用基于超声信号的光学检测方法,并专注于无接触式光声信号探测。
利用光外差干涉检测技术与数字多普勒信号解调方法,对非接触式光声信号探测展开了实验研究。通过对双频光外差干涉信号进行解调处理,成功地提取了被测目标的超声振动特征。采用超声换能器模拟高频振动信号,并对其进行性能测试。实验结果表明,外差系统能够较好地还原出超声振动信号,相对于水浸探头,解调出的信号频率误差相对较小。经验证,解调后可观察到纳米级的表面振动,所以光路系统的灵敏度为纳米量级,为进一步提高灵敏度,需要增加收集镜头,提高反射光收集能力。
此外,进行了碳棒和离体生物组织猪肝的光声信号探测实验。实验时,在样品前设置了测量镜M2,并采用耦合凝胶实现了M2与测量样本之间的耦合。这种反射镜的设置有助于收集背向散射光。在实际应用中可以在系统中去掉M2,通过增设信号光收集镜头来收集背向散射光。
最终实验结果显示,基于I/Q正交解调法的外差系统由于对干涉信号幅值变化不敏感,能够较好地解调出超声振动信号。同时,所设计的光声信号探测系统采用线偏振光外差干涉技术,提高了系统的检测灵敏度与精度。外差式光路结构易于调节,也能较好地抑制光源波动和环境光干扰,采用带通滤波可抑制载频外的低频噪声与高频噪声。利用高速数据采集卡和数字信号处理技术对干涉信号进行处理,解调算法简单,设计灵活且能节约硬件成本,对非接触式光声信号探测的临床应用具有一定的参考价值。
目前,该方法仍存在一定的局限性。在光声测量中,干涉光特别是信号光路的反射光强度直接影响测量的灵敏度和准确性,反射体的粗糙度、吸收特性、入射角度等都会影响干涉信号的幅值。在未来的研究中,需要进一步提高系统的灵敏度和精度,加强系统对散射光的收集,并优化信号处理方法。
[1] 李娇, 李帅, 陈冀景, 等. 非接触光声成像研究进展及其在生物医学上的应用[J]. 中国激光, 2021, 48(19): 1918005.
[2] Hosseinaee Z, Le M, Bell K, et al. Towards non-contact photoacoustic imaging [review][J]. Photoacoustics, 2020, 20: 100207.
[3] 万余洋, 雷鹏, 熊科迪, 等. 血管内光声-超声-光学相干层析-光声弹性多模态成像方法及系统[J]. 中国激光, 2023, 50(3): 0307107.
[4] 孟琪, 孙正, 侯英飒, 等. 校正光通量变化的定量光声内窥成像[J]. 光学学报, 2023, 43(1): 0111001.
[5] Chen Y W, Chen B H, Yu T F, et al. Photoacoustic mouse brain imaging using an optical Fabry-Pérot interferometric ultrasound sensor[J]. Frontiers in Neuroscience, 2021, 15: 672788.
[6] Deán-Ben X L, Pang G A, de Espinosa F M, et al. Non-contact optoacoustic imaging with focused air-coupled transducers[J]. Applied Physics Letters, 2015, 107(5): 051105.
[7] Sathiyamoorthy K, Strohm E M, Kolios M C. Low-power noncontact photoacoustic microscope for bioimaging applications[J]. Journal of Biomedical Optics, 2017, 22(4): 046001.
[8] Li H H, Cao F, Zhou Y Y, et al. Interferometry-free noncontact photoacoustic detection method based on speckle correlation change[J]. Optics Letters, 2019, 44(22): 5481-5484.
[9] Lu J, Gao Y Z, Ma Z H, et al. In vivo photoacoustic imaging of blood vessels using a homodyne interferometer with zero-crossing triggering[J]. Journal of Biomedical Optics, 2017, 22(3): 036002.
[10] Wang Y, Hu Y X, Peng B Y, et al. Complete-noncontact photoacoustic microscopy by detection of initial pressures using a 3×3 coupler-based fiber-optic interferometer[J]. Biomedical Optics Express, 2019, 11(1): 505-516.
[11] Eom J, Park S J, Lee B H. Noncontact photoacoustic tomography of in vivo chicken chorioallantoic membrane based on all-fiber heterodyne interferometry[J]. Journal of Biomedical Optics, 2015, 20(10): 106007.
[12] Eom J, Shin J G, Park S, et al. An all-fiber-optic combined system of noncontact photoacoustic tomography and optical coherence tomography[J]. Sensors, 2016, 16(5): 734.
[13] Bell K L, Hajireza P, Shi W, et al. Temporal evolution of low-coherence reflectrometry signals in photoacoustic remote sensing microscopy[J]. Applied Optics, 2017, 56(18): 5172-5181.
[14] Bell K, Hajireza P, Zemp R. Scattering cross-sectional modulation in photoacoustic remote sensing microscopy[J]. Optics Letters, 2018, 43(1): 146-149.
[15] Bell K L, Hajireza P, Zemp R J. Coherence-gated photoacoustic remote sensing microscopy[J]. Optics Express, 2018, 26(18): 23689-23704.
[16] Xu M, Wang L V. Photoacoustic imaging in biomedicine[J]. Review of Scientific Instruments, 2006, 77(4): 305-598.
[17] Yao D K, Zhang C, Maslov K, et al. Photoacoustic measurement of the Grüneisen parameter of tissue[J]. Journal of Biomedical Optics, 2014, 19(1): 017007.
[18] Petrova E, Ermilov S, Su R, et al. Using optoacoustic imaging for measuring the temperature dependence of Grüneisen parameter in optically absorbing solutions[J]. Optics Express, 2013, 21(21): 25077-25090.
[19] 陈重江, 周王婷, 胡毅成, 等. 非接触式光声成像技术及其应用[J]. 中国医疗设备, 2018, 33(1): 11-15.
Chen Z J, Zhou W T, Hu Y C, et al. Noncontact photoacoustic imaging and biomedical application[J]. China Medical Devices, 2018, 33(1): 11-15.
[20] 刘华锋. 光电检测技术及系统[M]. 2版. 杭州: 浙江大学出版社, 2020.
LiuH F. Optoelectric detection technology and system[M]. 2nd ed. Hangzhou: Zhejiang University Press, 2020.
[21] Yang P, Xing G Z, He L B. Calibration of high-frequency hydrophone up to 40 MHz by heterodyne interferometer[J]. Ultrasonics, 2014, 54(1): 402-407.
王成, 梁宸, 皇甫胜男, 朱俊, 张瑶, 徐锦程, 郑刚, 项华中, 张大伟. 基于激光外差干涉技术的非接触式光声信号检测研究[J]. 中国激光, 2024, 51(3): 0307402. Cheng Wang, Chen Liang, Shengnan Huangfu, Jun Zhu, Yao Zhang, Jincheng Xu, Gang Zheng, Huazhong Xiang, Dawei Zhang. Research on Non‑contact Photoacoustic Signal Detection Based on Laser Heterodyne Interferometry[J]. Chinese Journal of Lasers, 2024, 51(3): 0307402.