CoSi16-和Co2Si322-团簇几何结构、电子和热力学特性研究  下载: 726次
下载: 726次
1 引言
随着科学技术的高速发展,纳米团簇在新能源和新材料等方面表现出的优异物理化学特性引起了科学界的高度关注[1-5]。硅基纳米材料因具有独特的结构和优异的光学特性、电性能、磁特性和热导率等,在电子器件和纳米电子学等微电子工业中应用广泛[6-8]。硅原子位于第IVA族,由于其sp2轨道杂化的缺失,导致其存在大量悬挂键,使得硅团簇的稳定性很差。长期研究发现,一些金属或非金属原子掺杂硅团簇后,可以极大提高硅团簇的稳定性并产生诸多新颖的物理和化学特性[9-15]。其中,因外层d电子可以与硅团簇的悬挂键结合,大量的谱学实验和高精度理论计算广泛应用到过渡金属掺杂硅团簇的研究中[1,4,16-22]。其中,钴原子(3d74s2)掺杂硅团簇而产生的独特电学、磁性性质,更是理论和实验研究的热点[23-27]。例如,基于密度泛函理论下的B3LYP/6-311+G(d)方法,Fu等[23]对CoSin(n=6~9)团簇的几何结构演化和电子结构特性进行了深入研究。结果发现,CoSi9团簇为最小的笼状掺杂体系,CoSi8团簇由于存在诸多能量非常接近的能量异构体,导致该团簇的吸附活性降低。此外,钴原子通过spd杂化与周围的硅原子成键,进而提高了笼状结构的稳定性。基于实验的光电子能谱和量子化学的理论计算,Yang[24]的团队系统研究了CoSin-(n=3~12)团簇的几何结构和电子特性。结构演变发现,掺杂的Co原子在CoSi9-和CoSi10之后,从体系的外表面深入到体系的内部。磁性研究发现,除了CoSi3-和CoSi4-具有的总自旋磁矩为4 μB,其他团簇的磁性均为零。在研究范围内,高对称性(C3v)CoSi10-团簇具有相对高的稳定性。基于高精度的多组态二阶微扰理论(CASPT2),Tran等[25]系统研究了CoSin-/0/+(n=1~3)团簇的电子态。研究发现,BP86、PBE和TPSS泛函适合本体系对能量的计算。同时计算出了相应的组态、键长、振动频率和相对能量。最后,拟合出了CoSi3-团簇的光电子能谱。基于密度泛函理论的第一性原理计算,Robles和Khanna等[26]系统研究了CoSi2n团簇的结构和磁性。最近,Li等[22]基于实验和理论计算相结合,确定了中性CoSin(n=10~12)团簇的基态结构。随后,基于相同的方法,研究确定了CoSin+(n=5~8)和Co2Sin+(n=8~12)团簇的几何结构[27]。虽然广大科研工作者对钴掺杂硅团簇的结构和特性进行了一系列的研究,但是对中等尺寸钴掺杂硅团簇体系的研究较少。本文基于目前流行的结构预测方法——卡里普索结构预测方法及第一性原理计算,对CoSi16-团簇的结构进行了系统搜索,得到了CoSi16-团簇的基态和亚稳态结构。随后,基于CoSi16-团簇的基态结构,利用堆积的方式,在不同自旋下优化得到了二聚体Co2Si322-团簇的基态结构。基于上述基态结构,研究了两个体系的磁性和键级等电子特性。通过理论计算,拟合得到了两个体系的光电子能谱、红外和拉曼光谱。最后,分析了该体系的热力学特性,讨论了其热力学参数随温度变化的规律。
2 计算方法
本节利用吉林大学马琰铭教授课题组研发的卡里普索结构预测(CALYPSO)方法,对CoSi16-团簇结构进行搜索[28-30],该方法的合理性已被广泛验证[28-38]。该方法采用了基于对称性限制的随机结构产生技术、成键特征矩阵的结构相似性判断技术、粒子群优化算法的结构演化技术等来保证对团簇势能面的有效探索,可以合理地预测出研究体系的稳态结构。具体的结构搜索可以分为以下两步:第一步,通过设定搜索的代数和每代的步长,利用PBE0泛函和3-21G基组生成体系的1500个初始结构;第二步,从上述结构中抽取能量差超过0.3 eV的结构。此外,对于偶数个价电子的闭壳层体系,要考虑自旋多重度SM=1,3,5,7,
3 结果讨论
3.1 CoSi16-和Co2Si322-团簇结构
基于上节中的方法,优化得到了CoSi16-团簇的基态及亚稳态结构(
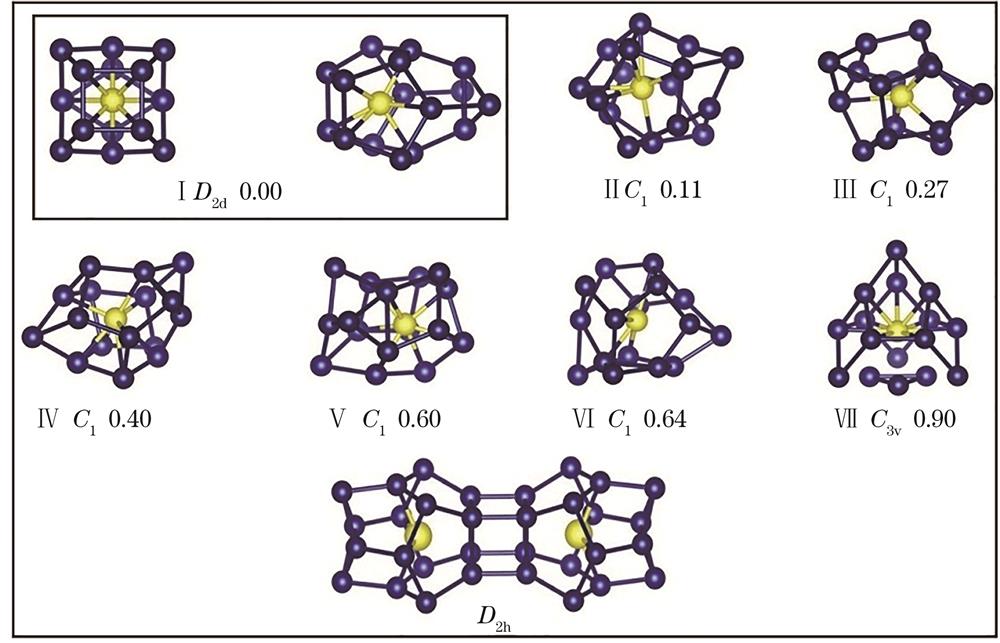
图 1. CoSi16-和Co2Si322-团簇的基态和亚稳态结构(Si原子:深色球体;Co原子:浅色球体。所有单位:eV)
Fig. 1. Lowest energy and low-lying structures of CoSi16- and Co2Si322- clusters (Si atoms: deep color spheres; Co atoms: light color spheres. All units: eV)
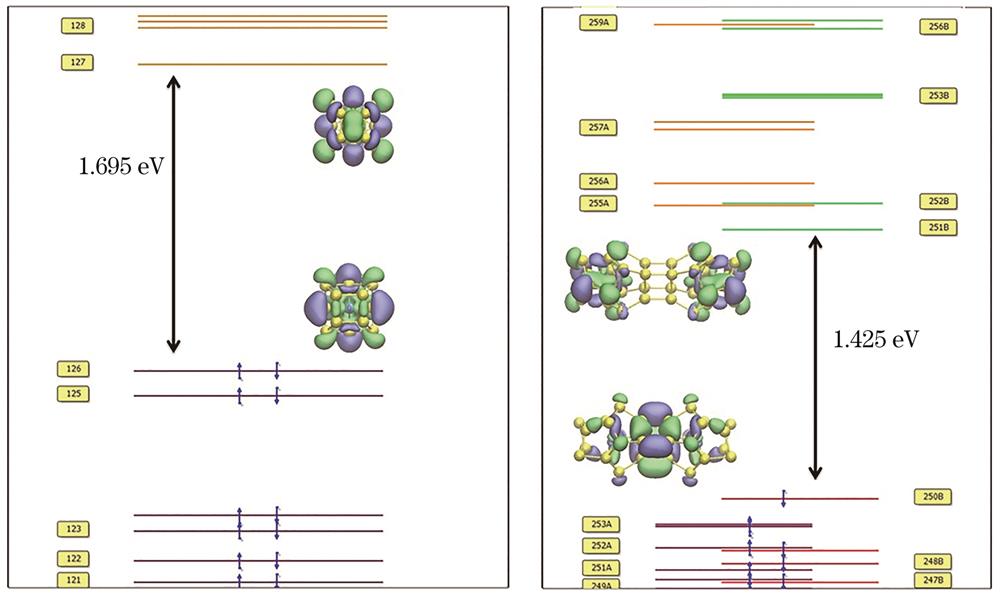
图 2. CoSi16-和Co2Si322-团簇基态结构对应的分子轨道能级图
Fig. 2. Molecular orbital energy levels of lowest energy structures of CoSi16- and Co2Si322- clusters
3.2 磁性分析
本节基于非限制性的UB3LYP泛函和6-311+G(d)全电子基组对团簇的磁学特性进行分析讨论。首先,对于CoSi16-团簇,理论分析发现该团簇总自旋磁矩为零,Co2Si322-团簇的总自旋磁矩为4 μB,堆积后团簇的磁性明显增强。
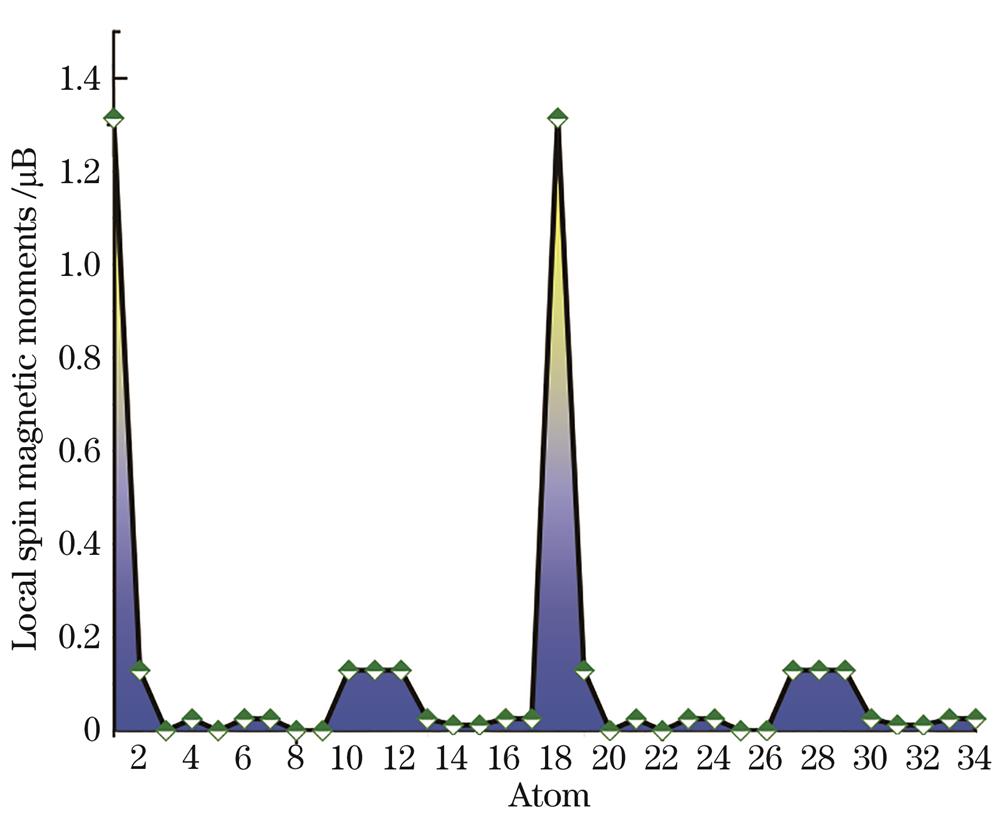
图 3. Co2Si322-团簇中各原子的局域自旋磁矩
Fig. 3. Local spin magnetic moments of atoms in Co2Si322- cluster
3.3 键级分析
为了探究CoSi16-和Co2Si322-团簇中不同原子之间相互作用的强弱,利用Multiwfn软件[42]计算得到了两个团簇中Si—Si和Co—Si的键长、Wiberg和FBO(fuzzy bond order)键级(见
表 1. CoSi16-和Co2Si322-团簇基态结构中Si—Si和Co—Si的键长和键级
Table 1. Bond lengths and bond orders of Si—Si and Co—Si of CoSi16- and Co2Si322- clusters
|
3.4 光电子能谱和红外、拉曼光谱
光电子能谱(PES)是实验上表征阴离子团簇的一种方法,能看作阴离子纳米团簇的电子指纹,它对结构非常敏感,可以通过对比理论和实验PES来验证体系的基态结构,PES为理论预测提供了方向性的指导,是探索团簇新颖结构与性质的重要手段。本节利用Multiwfn软件,基于Koopmans定理[43],轨道的相对能量按公式ΔE=E(HOMO-n)-E(HOMO)计算所得,第一个峰值对应垂直解离能(VDE),拟合时所有的峰均采用0.2 eV高斯展宽进行模拟,横坐标为电子能量,纵坐标是光电子强度[
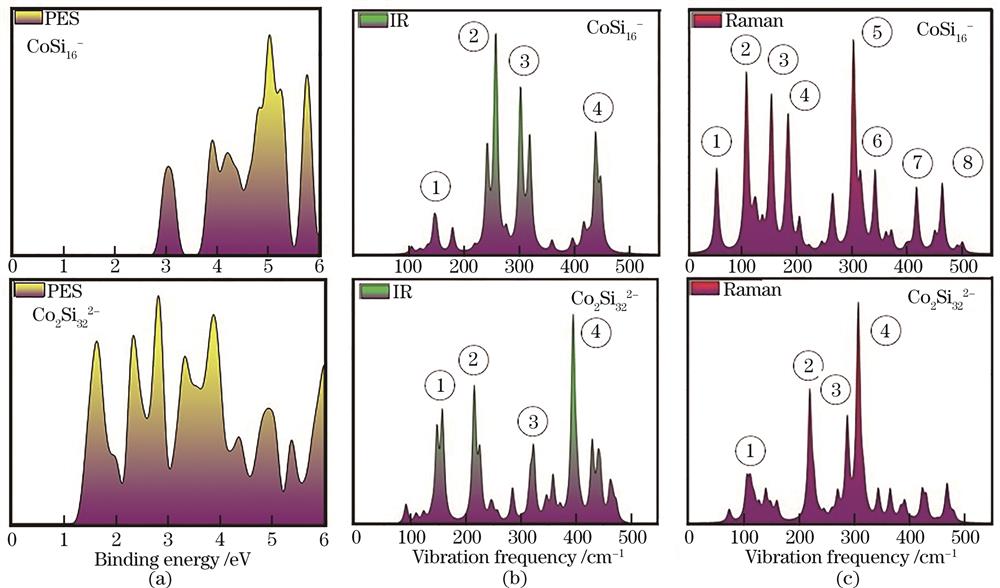
图 4. 拟合CoSi16-和Co2Si322-团簇的(a)PES、(b)IR和(c)Raman光谱
Fig. 4. Simulated (a) PES, (b) IR, and (c) Raman spectra of CoSi16- and Co2Si322- clusters
基于B3LYP泛函和6-311+G(d)基组,图
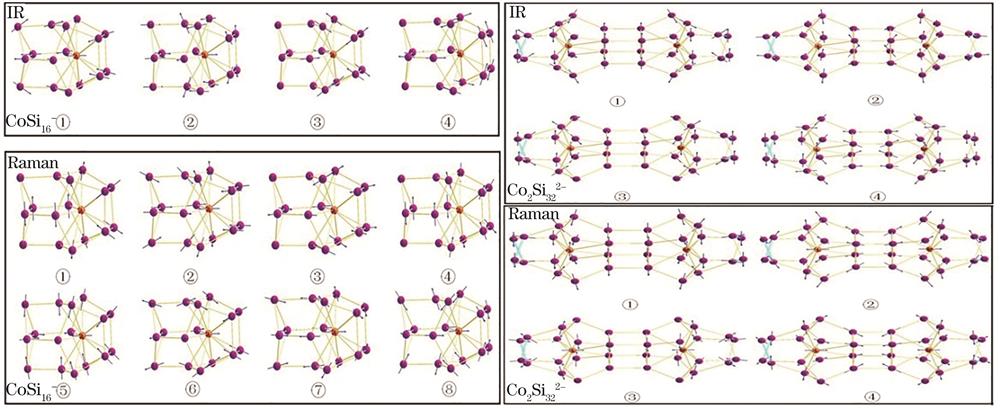
图 5. CoSi16-和Co2Si322-团簇中特征峰对应原子的振动方向
Fig. 5. Vibration direction of atoms in active peaks for CoSi16- and Co2Si322- clusters
3.5 热力学特性
在温度为298.15 K,大气压为1.013 ×105 Pa时,利用B3LYP/6-311+G(d)方法计算得到了两个体系的热力学参数:定容热容Cv、标准熵S和标准生成焓
式中计算结果见
表 2. CoSi16-和Co2Si322-团簇基态结构对应的热力学参数
Table 2. Calculated thermodynamic parameters of CoSi16- and Co2Si322- clusters
|
表 3. 不同温度下CoSi16-和Co2Si322-团簇的热力学参数
Table 3. Thermodynamic parameters at different temperatures of CoSi16- and Co2Si322- clusters
|
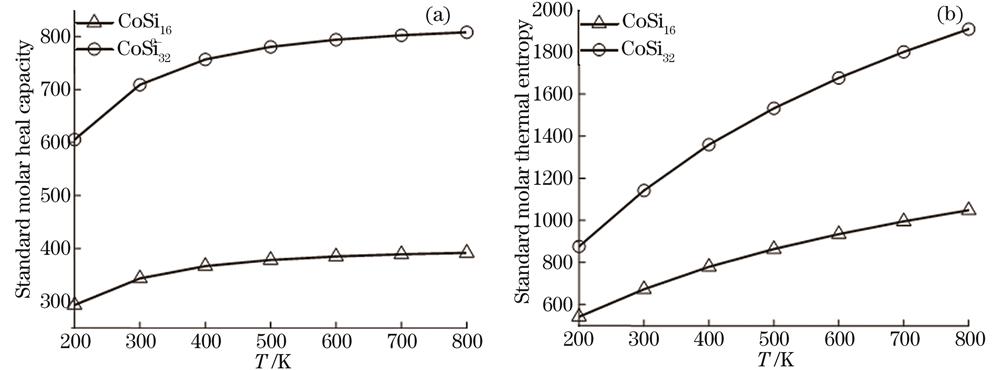
图 6. CoSi16-和Co2Si322-团簇的热力学参数(a)Cv和(b)S随温度变化曲线
Fig. 6. Temperature dependence of (a) Cv and (b) S for CoSi16- and Co2Si322- clusters
从
对应的测定系数R2分别为0.9706、0.9703、0.9993和0.9993。
4 结论
基于卡利普索结构预测方法和密度泛函理论的第一性原理计算,对CoSi16-和Co2Si322-团簇的几何结构、电子和热力学特性进行了系统研究。结果发现,CoSi16-团簇的基态具有高对称性D2d点群对称结构。以此结构为基地,通过堆积优化后的二聚体Co2Si322-具有高对称的D2h点群对称结构。磁性分析发现,CoSi16-团簇不具有磁性,Co2Si322-团簇具有的总自旋磁矩为4 μB。其中,Co原子对总自旋磁矩的贡献大,Si原子贡献较小,部分Si原子对总自旋磁矩的贡献为零。键长和能级分析表明,两个体系中Co—Si原子间的相互作用强于Si—Si原子间的相互作用,CoSi16-团簇的稳定性要强于Co2Si322-团簇的稳定性。此外,两体系的IR和Raman谱的特征峰多对应呼吸振动的模式,而且,IR和Raman谱的振动模式都具有较强的对称性。最后,热力学性质分析表明,两体系在热力学上都是稳定的;随着堆积团簇尺寸的增大,定容热容和标准熵随之增大。
[1] 刘振平, 姜容, 庞钶靖. 还原型谷胱甘肽修饰的金纳米团簇荧光传感器对铜离子的高灵敏检测[J]. 激光与光电子学进展, 2021, 58(14): 1404002.
[3] 李成刚, 张洁, 申梓刚, 等. NiB20-团簇的结构、电子和光谱特性研究[J]. 光学学报, 2020, 40(20): 2016001.
[6] Holmes J D, Johnston K P, Doty R C, et al. Control of thickness and orientation of solution-grown silicon nanowires[J]. Science, 2000, 287(5457): 1471-1473.
[7] Haertelt M, Lyon J T, Claes P, et al. Gas-phase structures of neutral silicon clusters[J]. The Journal of Chemical Physics, 2012, 136(6): 064301.
[8] Vogel M, Kasigkeit C, Hirsch K, et al. 2p core-level binding energies of size-selected free silicon clusters: chemical shifts and cluster structure[J]. Physical Review B, 2012, 85(19): 195454.
[9] Lu S J. Structural evolution from exohedral to endohedral geometries, dynamical fluxionality, and structural forms of medium-sized anionic and neutral Au2Sin (n = 8-20) clusters[J]. Physical Chemistry Chemical Physics, 2020, 22(44): 25606-25617.
[10] Chen Z H, Ai L N, Li Y X, et al. Density functional theory investigation on cluster assembled Gd-encapsulated Si nanowires[J]. Physics Letters A, 2020, 384(33): 126848.
[11] Li C G, Chen W G, Cui Y Q, et al. Structures, stabilities and electronic properties of the bimetal V2-doped Sin (n=1-10) clusters: a density functional investigation[J]. The European Physical Journal D, 2020, 74: 111.
[12] Lu S J, Xu X L, Xu H G, et al. Structural evolution and bonding properties of Au2Sin-/0 (n = 1-7) clusters: Anion photoelectron spectroscopy and theoretical calculations[J]. The Journal of Chemical Physics, 2018, 148(24): 244306.
[13] Zhao Y R, Bai T T, Jia L N, et al. Probing the structural and electronic properties of neutral and anionic lanthanum-doped silicon clusters[J]. The Journal of Physical Chemistry C, 2019, 123(47): 28561-28568.
[14] Bae G T. Density functional theory calculations of geometrical and electronic properties of neutral and charged silicon oxide clusters[J]. Bulletin of the Korean Chemical Society, 2019, 40(8): 780-786.
[15] Pham H T, Majumdar D, Leszczynski J, et al. 4d and 5d bimetal doped tubular silicon clusters Si12M2 with M = Nb, Ta, Mo and W: a bimetallic configuration model[J]. Physical Chemistry Chemical Physics: PCCP, 2017, 19(4): 3115-3124.
[16] Claes P, Janssens E, Ngan V T, et al. Structural identification of caged vanadium doped silicon clusters[J]. Physical Review Letters, 2011, 107(17): 173401.
[17] Wang J G, Zhao J J, Ma L, et al. Stability and magnetic properties of Fe encapsulating in silicon nanotubes[J]. Nanotechnology, 2007, 18(23): 235705.
[18] Janssens E, Lievens P. Growth mechanisms for doped clusters[J]. Advances in Natural Sciences: Nanoscience and Nanotechnology, 2011, 2(2): 023001.
[19] Zheng W J, Nilles J M, Radisic D, et al. Photoelectron spectroscopy of chromium-doped silicon cluster anions[J]. The Journal of Chemical Physics, 2005, 122(7): 071101.
[20] Claes P, Ngan V T, Haertelt M, et al. The structures of neutral transition metal doped silicon clusters, SinX (n = 6-9; X = V, Mn)[J]. The Journal of Chemical Physics, 2013, 138(19): 194301.
[21] Koyasu K, Atobe J, Akutsu M, et al. Electronic and geometric stabilities of clusters with transition metal encapsulated by silicon[J]. The Journal of Physical Chemistry A, 2007, 111(1): 42-49.
[22] Li Y J, Tam N M, Claes P, et al. Structure assignment, electronic properties, and magnetism quenching of endohedrally doped neutral silicon clusters, SinCo (n = 10-12)[J]. The Journal of Physical Chemistry A, 2014, 118(37): 8198-8203.
[23] 傅院霞, 王倩, 吕思斌, 等. 过渡金属原子钴掺杂硅团簇CoSi6~9的密度泛函理论研究[J]. 原子与分子物理学报, 2011, 28(5): 859-865.
Fu Y X, Wang Q, Lü S B, et al. Density functional theory investigation on Co-doped silicon clusters, CoSin(n=6-9)[J]. Journal of Atomic and Molecular Physics, 2011, 28(5): 859-865.
[24] Yang B, Xu X L, Xu H G, et al. Structural evolution and electronic properties of CoSin- (n = 3-12) clusters: mass-selected anion photoelectron spectroscopy and quantum chemistry calculations[J]. Physical Chemistry Chemical Physics: PCCP, 2019, 21(11): 6207-6215.
[25] Tran V T. Electronic states of CoSin–/0/+ (n = 1-3) clusters from density matrix renormalization group-CASPT2 calculations[J]. The Journal of Physical Chemistry A, 2021, 125(26): 5800-5810.
[26] Robles R, Khanna S N. Stable T2Sin (T=Fe, Co, Ni, 1≤n≤8) cluster motifs[J]. The Journal of Chemical Physics, 2009, 130(16): 164313.
[27] Li Y J, Tam N M, Woodham A P, et al. Structure dependent magnetic coupling in cobalt-doped silicon clusters[J]. The Journal of Physical Chemistry C, 2016, 120(34): 19454-19460.
[28] Wang Y C, Lü J, Zhu L, et al. Crystal structure prediction via particle-swarm optimization[J]. Physical Review B, 2010, 82(9): 094116.
[29] Wang Y C, Lü J, Zhu L, et al. CALYPSO: a method for crystal structure prediction[J]. Computer Physics Communications, 2012, 183(10): 2063-2070.
[30] Wang Y C, Miao M S, Lü J, et al. An effective structure prediction method for layered materials based on 2D particle swarm optimization algorithm[J]. The Journal of Chemical Physics, 2012, 137(22): 224108.
[31] Lu C, Miao M S, Ma Y M. Structural evolution of carbon dioxide under high pressure[J]. Journal of the American Chemical Society, 2013, 135(38): 14167-14171.
[32] Zhao Y R, Xu Y Q, Chen P, et al. Structural and electronic properties of medium-sized beryllium doped magnesium BeMgn clusters and their anions[J]. Results in Physics, 2021, 26: 104341.
[33] Li C G, Li H J, Cui Y Q, et al. A density functional investigation on the structures, electronic, spectral and fluxional properties of VB20-cluster[J]. Journal of Molecular Liquids, 2021, 339: 116764.
[34] Jin S Y, Chen B L, Kuang X Y, et al. Structural and electronic properties of medium-sized aluminum-doped boron clusters AlBn and their anions[J]. The Journal of Physical Chemistry C, 2019, 123(10): 6276-6283.
[35] Pang J F, Jin W Y, Kuang X Y, et al. Two-dimensional Fe8N nanosheets: ferromagnets and nitrogen diffusion[J]. The Journal of Physical Chemistry Letters, 2021, 12(35): 8453-8459.
[36] Chen B L, Sun W G, Kuang X Y, et al. Structural stability and evolution of medium-sized tantalum-doped boron clusters: a half-sandwich-structured TaB12- cluster[J]. Inorganic Chemistry, 2018, 57(1): 343-350.
[37] Li C G, Shen Z G, Zhang J, et al. Analysis of the structures, stabilities and electronic properties of MB16- (M = V, Cr, Mn, Fe, Co, Ni) clusters and assemblies[J]. New Journal of Chemistry, 2020, 44(13): 5109-5119.
[38] Lu C, Gong W G, Li Q, et al. Elucidating stress–strain relations of ZrB12 from first-principles studies[J]. The Journal of Physical Chemistry Letters, 2020, 11(21): 9165-9170.
[39] Becke A D. Density-functional thermochemistry. III. The role of exact exchange[J]. The Journal of Chemical Physics, 1993, 98(7): 5648-5652.
[40] Krishnan R, Binkley J S, Seeger R, et al. Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions[J]. The Journal of Chemical Physics, 1980, 72(1): 650-654.
[41] FrishM J, TrucksG W, SchlegelH B, et al. Gaussian 09[M]. Wallingford, CT: Gaussian Inc., 2009.
[42] Lu T, Chen F W. Multiwfn: a multifunctional wavefunction analyzer[J]. Journal of Computational Chemistry, 2012, 33(5): 580-592.
[43] Tozer D J, Handy N C. Improving virtual Kohn-Sham orbitals and eigenvalues: application to excitation energies and static polarizabilities[J]. The Journal of Chemical Physics, 1998, 109(23): 10180-10189.
[44] Zhang S, Dai W, Liu H Z, et al. Geometrical and electronic structure of the Ba-doped Sin (n = 1-12) cluster: a density functional study[J]. Journal of Molecular Structure, 2014, 1075: 220-226.
Article Outline
李家秀, 郭江水, 程琳, 郭梦姣, 王子昂, 李成刚, 崔颍琦. CoSi16-和Co2Si322-团簇几何结构、电子和热力学特性研究[J]. 激光与光电子学进展, 2023, 60(1): 0102001. Jiaxiu Li, Jiangshui Guo, Lin Cheng, Mengjiao Guo, Ziang Wang, Chenggang Li, Yingqi Cui. Study on Geometric Structures and Electronic and Thermodynamic Properties of CoSi16- and Co2Si322- Clusters[J]. Laser & Optoelectronics Progress, 2023, 60(1): 0102001.






